Acute promyelocytic leukemia (APL) is a hematological emergency associated with high early mortality rates, thus requiring prompt diagnosis and treatment initiation. Currently, diagnostic suspicion relies on morphological criteria, which can be further corroborated by immunophenotypic analyses, until results based on molecular techniques confirm the diagnosis. However, while both cytology and immunophenotypic analyses are very important in this setting, their accuracy can be influenced by inherent morphological variation of blasts, as well as by gating and other technical issues related to flow cytometry. In this context, novel parameters that could contribute to the segregation of APL from other forms of acute myeloid leukemia (AML) would be desirable. Recently, Lavallée et al demonstrated that the aberrant expression of podoplanin (PDPN), a protein that mediates platelet activation in inflammatory contexts, by leukemic cells is as a relevant mediator of the coagulopathy of APL, as well potential biomarker of this condition based on the analysis of primary cells from patients with AML.
The aim of this study was to prospectively and independently validate whether the surface expression of PDPN by leukemic promyelocytes measured by flow cytometry can contribute to the differential diagnosis between APL and other forms of AML. We also explored the association of PDPN expression with clinical and laboratory characteristics of these patients.
The study population consisted of all consecutive patients diagnosed with APL by cytological, immunophenotypical and molecular techniques in our adult academic hematology hospital from 2020 to 2023. Exclusion criteria included the presence of hemodynamic instability or refusal to participate. The expression of PDPN was measured by flow cytometry using BD FACSCanto II™ cytometer. Analysis was performed in accordance with EuroFlow protocols, with a panel that includes CD45, CD33, CD34, MPO, CD117, CD13, HLA-DR, as well as an antibody against PDPN (PE anti-human Podoplanin Antibody - Catalog# 337004 - BioLegend). Lymphocytes were used as internal negative control for PDPN expression. Data were analyzed with Infinicyt™ software and reported both as the % of PDPN + cells or according to mean fluorescence intensity (MFI). Patients with other forms of AML diagnosed during the same period and matched for age, gender and peripheral blast count were used to compare the diagnostic accuracy of PDPN measurement. Clinical and laboratory data were obtained from the medical records. The study was approved by the Institutional Review Board and all participants provided written informed consent.
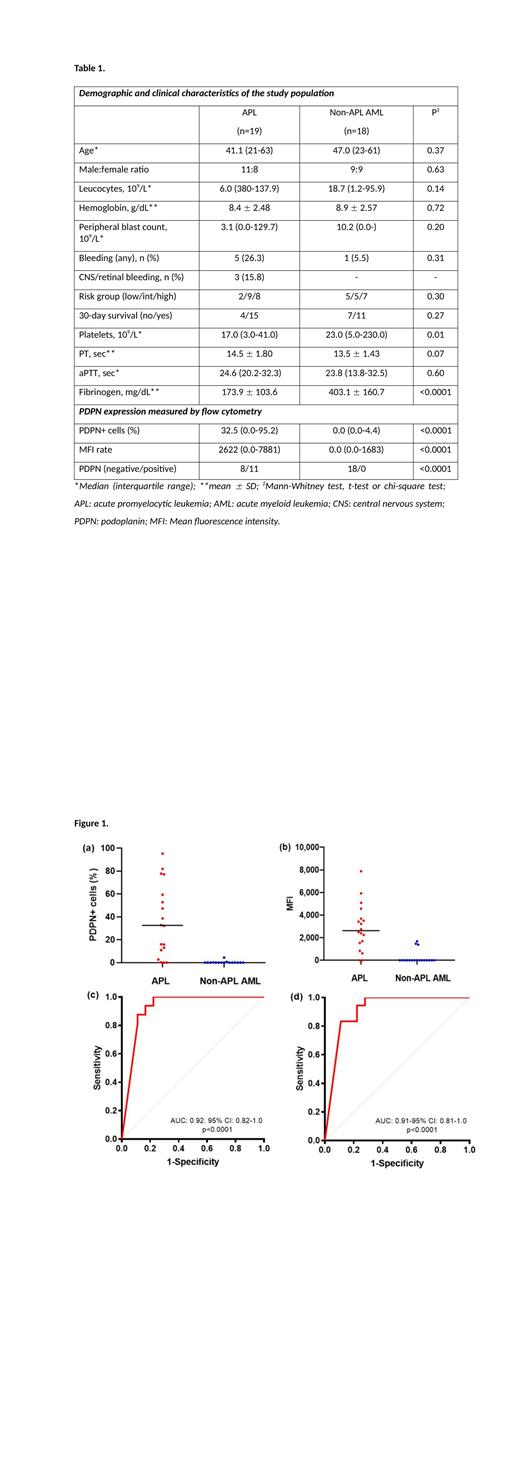
In total, 19 consecutive patients with confirmed diagnosis of APL were compared with 18 patients with non-APL AML. Clinical and laboratory data from the study population are shown in table 1. The expression of PDPN in leukemic blasts was significantly higher in APL compared to non-APL AML, considering either the percentage of PDPN + cells or the MFI (table 1; figure 1a-b). When considered as a categorical variable, the expression of PDPN was strongly associated with the diagnosis of APL (Chi-square test= 19.169; P<0.001). PDPN expression was inversely correlated with platelet count (R S= -0.47; P=0.003) and fibrinogen (R S= -0.51; P=0.001), but not with other laboratory parameters such as PT and aPTT. No association of PDPN expression with the available clinical variables was observed, although it should be noted that our study is underpowered to confirm these findings. We next estimated the diagnostic accuracy of PDPN expression by flow cytometry by ROC analysis. As shown in figures 1c-d, the area under the ROC curve was higher than 0.9 for both analytical strategies.
In conclusion, our prospective study demonstrated that measurement of the expression of PDPN by flow cytometry at diagnosis is capable to accurately segregate APL from other forms of AML. Additional studies are warranted to explore the clinical association of PDPN expression in larger and independent cohorts.
Disclosures
Saraiva:Janssen Global Services, LLC: Speakers Bureau; AstraZeneca: Speakers Bureau; akeda Pharmaceutical Company Limited: Speakers Bureau. Duarte:Astrazeneca: Speakers Bureau. Oliveira:Janssen: Speakers Bureau. Saad:FAPESP Sao Paulo state foundation: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal